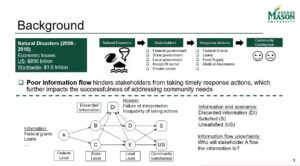
Today, Virginia is one step closer to bridging the gap between science and policy — a critical relationship necessary to navigate a range of socio-technical issues including climate change, biodiversity loss, pandemics, and poverty. Creating this connection was the result of a yearlong research project to identify U.S. and Virginia science policy programs which engage STEM-H scientists and engineers in science policy with the end result of helping both elected and non-elected officials access technical information to assist in decision making.


This work was completed thanks to a coalition of faculty and students supported though a 4-VA@Mason grant. The team was led by KL Akerlof in George Mason’s Department of Environmental Science & Policy and Todd Schenk, Chair and Associate Professor in the Urban Affairs and Planning Program of the School of Public and International Affairs at Virginia Tech and Director of the Science, Technology & Engineering in Policy Program.
To advance best practices for professional mentorship of early career researchers and to build capacity for training researchers to engage in public policy, the team developed a database of U.S. science policy programs and conducted a case study of those in Virginia through surveys and interviews with their leaders. Akerlof and Schenk were assisted in the study by Adriana Bankston, an expert in science policy, and Mason students Kelsey Mitchell, Kate Saylor, and Aniyah Syl, and Kenneth Dewberry at Virginia Tech.
Following the yearlong collaboration, the results—along with program blogs and a listserv—has been developed and is featured on a new website created to support this emerging network of science policy programs: https://scipolprograms.org/.

The results of their research showed that the majority (57%) of U.S. science policy programs are state based. These programs include student organizations, government placements and fellowships, and academic certificates, degrees, and other training. However, it was recognized that there is only a limited ability to implement evidence-based approaches within these programs.
The team compared programmatic outcomes from Virginia’s science policy programs with those described in academic literature. Academic and professional scholarship in science communication, and public affairs suggests that curricula for engaging scientists and engineers should broadly cover communication and policy processes.
In addition, they found that training programs would benefit from evaluation models and measures, although the lack of consistent theoretical foundations and constructs across this highly multidisciplinary scholarship reduces their utility. It was concluded that a common framework, which includes shared terms and relationships, is needed to promote the transdisciplinary growth of the field.
With the results of their analysis in hand, the team held a state webinar on science policy programs featuring 11 of the 13 Virginia program leaders. The project was also highlighted at a Virginia Sea Grant Symposium and was the topic of an American Geophysical Union e-lightning talk.

While the results of the study proved important, the project also provided rich opportunities to the students participating in the effort. Undergraduate student Syl and doctoral candidate Mitchell helped lead the webinar and participated as co-authors on the publications. Further, Syl co-presented at the Virginia Sea Grant Symposium. Plus, master’s student Saylor successfully defended her thesis on the Commonwealth of Virginia Engineering and Science (COVES) Policy Fellowship. She subsequently presented her results to the President of the Virginia Academy of Science, Engineering, and Medicine, Dr. James Aylor.
The first of two research articles based on the project has just been accepted with minor revisions by the journal Evidence & Policy. A second article has also been submitted for publication. They are also in conversation with the leadership of the Virginia Academy of Science, Engineering, and Medicine about the development of a new weeklong science policy program for undergraduate students from across the Commonwealth’s public universities.
Akerlof reflects on receiving the 4-VA@Mason award, “This funding supported our cross-institutional collaboration and ability to conduct baseline research and networking that have been fundamental to understanding how the landscape of science policy training programs is evolving across the United States.”



 Professor in George Mason’s College of Science, Department of Biology, saw an opportunity to combine the experience and knowledge of other experts in the field to develop a novel genomic tool to address this issue.
Professor in George Mason’s College of Science, Department of Biology, saw an opportunity to combine the experience and knowledge of other experts in the field to develop a novel genomic tool to address this issue. remain In the wild, there are approximately 160 bongos in U.S. zoos and several hundred more on private ranches. As such, breeding and transfer strategies that increase genetic diversity and reduce inbreeding of captive animals are critical for the survival of the species.
remain In the wild, there are approximately 160 bongos in U.S. zoos and several hundred more on private ranches. As such, breeding and transfer strategies that increase genetic diversity and reduce inbreeding of captive animals are critical for the survival of the species. GMU researchers, Lim and his PhD student, Karen Holm, assembled their team — Aakrosh Ratan, Assistant Professor, University of Virginia; Klaus-Peter Koepfli, Senior Research Scientist, Smithsonian Mason School of Conservation; and Budhan Pukazhenthi, Research Physiologist, Smithsonian Conservation Biology Institute. 4VA funding was used to purchase the necessary supplies, reagents and equipment including DNA tests and probe sets.
GMU researchers, Lim and his PhD student, Karen Holm, assembled their team — Aakrosh Ratan, Assistant Professor, University of Virginia; Klaus-Peter Koepfli, Senior Research Scientist, Smithsonian Mason School of Conservation; and Budhan Pukazhenthi, Research Physiologist, Smithsonian Conservation Biology Institute. 4VA funding was used to purchase the necessary supplies, reagents and equipment including DNA tests and probe sets. centers and zoos were collected to begin the project. This resulted in a total of 207 samples from both current and historical ex situ populations (populations outside of the native ranges). They evaluated the effectiveness of the probe-set in generating high-quality genetic data from different sample types and for different genetic marker types. The data obtained are now being accessed for their ability to produce accurate information on animal kinship, genetic diversity and population structure, which will ultimately be used to guide precise captive breeding programs.
centers and zoos were collected to begin the project. This resulted in a total of 207 samples from both current and historical ex situ populations (populations outside of the native ranges). They evaluated the effectiveness of the probe-set in generating high-quality genetic data from different sample types and for different genetic marker types. The data obtained are now being accessed for their ability to produce accurate information on animal kinship, genetic diversity and population structure, which will ultimately be used to guide precise captive breeding programs.
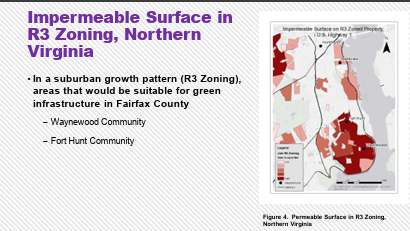
 That was just the challenge Younsung Kim in Mason’s Department of Environmental Science and Policy wanted to take on. Kim, who has an extensive background in complex environmental and sustainability issues, saw the potential of incorporating freely available data from US Geological Society and Census Bureau along with the local county government land use zoning data to create a computational spatial model to help identify what works, and what doesn’t work, in GI.
That was just the challenge Younsung Kim in Mason’s Department of Environmental Science and Policy wanted to take on. Kim, who has an extensive background in complex environmental and sustainability issues, saw the potential of incorporating freely available data from US Geological Society and Census Bureau along with the local county government land use zoning data to create a computational spatial model to help identify what works, and what doesn’t work, in GI.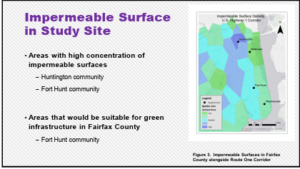 Finally, Kim wanted to incorporate today’s trends in GI architecture in the research; identifying UVA’s School of Architecture as a leading source of urban planning and spatial analysis expertise.
Finally, Kim wanted to incorporate today’s trends in GI architecture in the research; identifying UVA’s School of Architecture as a leading source of urban planning and spatial analysis expertise. Political Science Association Conference, and at a Mason Earth Day event. A paper entitled “Mapping Green Infrastructure from Stormwater” was published in Environmental Pollution and Climate Change.
Political Science Association Conference, and at a Mason Earth Day event. A paper entitled “Mapping Green Infrastructure from Stormwater” was published in Environmental Pollution and Climate Change.